The United States Food and Drug Administration has approved the marketing of a prosthetic arm nicknamed “Luke” in a nod to the bionic hand grafted onto Star Wars hero Luke Skywalker after his was cut off by Darth Vader’s light saber.
The arm, eight years in the making, was funded by the military.
It was developed by Deka Research, founded by Dean Kamen, the inventor of the Segway.

“We are happy the FDA has approved the Deka Arm for commercialization,” Tom Doyon, Deka’s project manager for the device, told TechNewsWorld. “This will let us focus on manufacturing and bringing to market this system.”
Statistics on Arm Loss
There are 350,000 amputees in the U.S., according to ISHN. Of these, 30 percent — more than 115,000 — have lost an upper limb. Of the Americans who have lost part or all of an upper limb, about 10 percent have lost their wrist and hand.
Reasons for amputation include cardiovascular disease, trauma, infection and nerve injury. Some people are missing limbs due to congenital anomalies.
U.S. military statistics show 1,558 troops lost one or more limbs or partial limbs, or one or more full or partial hands or feet, in Iraq and Afghanistan from 2001 to 2013.
People often do not use their prosthetic limbs for a variety of reasons, Troy Turner, member of the Amputee Coalition’s Scientific and Medical Advisory Committee, told TechNewsWorld.
“Many amputees learn how to do things with one arm rather than endure the pain, discomfort or hassle of a prosthetic device,” Turner said. “An arm such as [the Luke] may overcome these factors and bring significantly more usability to the wearer.”
What’s in the Luke
The Luke, or more properly the Deka Arm System, is the first prosthetic arm that can perform multiple simultaneous powered movements controlled by electrical signals from electromyogram electrodes.
Typically, an upper extremity prosthesis is controlled by using muscle contractions that are not the same as those used to control a biological arm, Turner said.
The electrodes in the Deka arm send electrical signals caused by muscle contractions close to where the prosthesis is attached to a computer processor in the prosthesis that then translates them into up to 10 specific movements.
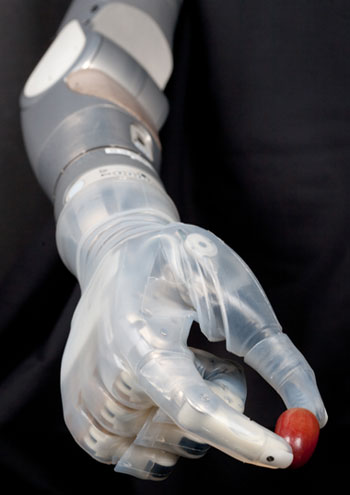
“It’s all custom electronics,” Doyon said. He declined to specify which processors are used.
The Deka arm also has switches, movement sensors and force sensors that cause it to move.
Come Here, Skywalker
The Deka arm allows for simultaneous control of multiple joints using a variety of input devices including wireless signals generated by innovative sensors on the user’s feet, DARPA said.
The arm is configurable for different levels of limb loss — between the elbow and the wrist, from the shoulder to the elbow, or from the shoulder itself, Deka’s Doyon said.
It was tested on 36 subjects at the U.S. Veterans Administration between 2009 and 2012.
The FDA’s Role
The FDA reviewed various reports arising out of the tests conducted with the arm.
Follow-up monitoring will be required.
“Given this is a completely novel technology with complex machinery, there will be a significant user learning curve, and it’s likely ongoing support for tuneups or other adjustments to ensure proper functionality would be needed,” Venkat Rajan , advanced medical technologies principal analyst at Frost & Sullivan, told TechNewsWorld.
“The FDA uses Medical Device Reporting as a post-market surveillance tool to monitor device performance, detect potential device-related safety issues, and contribute to benefit-risk assessments of these products,” agency spokesperson Susan Laine told TechNewsWorld.
Who Can Get One?
It’s not yet clear where and when the device will be available. Deka’s Doyon could not say.
“This is an exciting and groundbreaking technology that gives new options to patients,” Rajan noted. “The remaining question is price. To what degree will it be covered by insurers? Or will it require out-of-pocket expenditure by the individual?”