Apple is working on a secret project to develop wearable devices that can monitor the blood sugar of diabetics without using invasive finger sticks, part of a vision that originated with company founder Steve Jobs, CNBC reported earlier this week.
Apple has assembled a team of biomedical engineers from various companies to work on the project, according to the report.
Cor, a company Apple acquired in 2010, has been working for more than five years on a way to integrate noninvasive glucose monitoring into a wearable like an Apple Watch device.
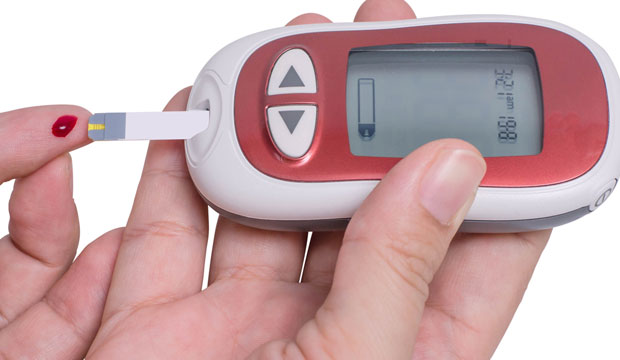
Glucose monitoring traditionally has required that diabetics use lancets to pierce their fingertips at least four times daily, measuring blood glucose levels before and after meals, when waking up, and before going to bed.
Many Type 1 diabetics wear pumps to deliver insulin, and they sometimes test up to 16 times a day.
One of the benefits of continuous glucose monitoring, or CGM, is to warn diabetics when blood glucose is rising or falling rapidly. Hypoglycemia can result when glucose levels fall below 70 milligrams per deciliter. Hyperglycemia, a rapid rise of blood glucose, can lead to ketoacidosis or, worse, a diabetic coma.
Apple reportedly has begun feasibility studies in the Bay Area and has retained consultants to help with regulatory issues.
Lucrative Market
Managing diabetes has been a rapidly evolving focus in both the smartphone and wearable device markets.
Integrating glucose sensors into wearable devices is a difficult challenge, but the market potential is very great, noted Jitesh Ubrani, senior research analyst for WW mobile device trackers at IDC.
“Today most wearables are consumer products, but there’s an untapped opportunity in the medical community,” he told TechNewsWorld.
The overall diabetic testing market will reach US$17 billion in 2021, up from $12 billion in 2016, ABI has estimated. Revenue from CGM devices is expected to increase at a compound annual growth rate of 41 percent. More than 9 million wearable CGM devices are expected to ship by 2021, and wearable device makers like Apple and other competitors want to get a bite of that market.
Existing Products
Apple has an existing partnership with Dexcom, which offers the G5 continuous glucose monitor. It allows Apple Watch users to monitor their blood sugar using an app that syncs with iPhone.
Many CGM devices have accuracy issues and require frequent calibration with blood glucose sensors. However, regulators last year found the G5 readings were accurate enough to require calibration just once every 12 hours.
Abbott’s Libre blood testing system has a sensor worn on the upper arm up to 14 days at a time, which measures interstitial fluid under the skin every minute. The system originated in 2014 in Europe and now is available in 30 countries worldwide; it is under review by the U.S. Food and Drug Administration.
Medtronic, one of the leading makers of diabetic insulin pumps and continuous glucose sensors in the U.S. last year received approval in Europe, Latin America and Australia for its Guardian Connect device, according to company spokesperson Danielle Swanson. The device currently is under review by the U.S. Food and Drug Administration.
“CGM is extremely valuable to people with diabetes, as it allows them to see current glucose levels at any time on their phone without a fingerstick,” Swanson told TechNewsWorld. “People with diabetes can also receive alerts to help them avoid high and low blood glucose levels.”
The app, which is being rolled out on a country by country basis, initially was made for the iOS system. Medtronic will release an Android version at a later date.
Under a partnership announced last year, Medtronic will offer glucose tracking on Fitbit’s iPro2 mLog mobile app, using data from Medtronic’s iPro2 professional CGM system, to give patients a record of their exercise and blood glucose levels. The app is available for both iOS and Android mobile devices.
UK-based Nemaura Medical has developed the sugarBEAT system, a glucose monitoring device that uses a noninvasive, disposable patch. The 10mm coin-sized device measures molecular amounts of interstitial fluid using an electric current .
The same technology is being developed for a variety of other uses, such as taking measurements related to athletic performance and measuring oxygen depletion in patients. The patch would last up to 14 days before needing to be changed.
The company signed a letter of intent with a China-based Shenzen CAS Health Corp. in a joint venture to manufacture and distribute the sensors in China, pending regulatory approval.
“We have designed sugarBEAT around the needs of the user,” Nemaura Medical Director Bashir Timol told TechNewsWorld. “Accordingly, the primary interface is the app accessible on users’ existing smart device.”





















































I AM all for anything that makes the management of Type 1 Diabetes easier. But as the previous comment stated, pricking the finger is minor compared to all the other things that are associated with the disease. The article talks about the multi-billion market that is out there, that Apple wants to be a part of. I AM also all for making a buck, but I really don’t care for the greed that drips from that statement. I, and millions of others with this silent killer disease, would love to see a cure! On the other hand, what would happen to all these companies, i.e. Lilly, Medtronic, Apple, etc, etc, if the cure was found? Granted, I know that they have fingers in other financial pots, but if by 2021, the CGM market alone will reach $17B, that is a huge chunk of money being said good-bye to. I AM not a conspiracy theorist, but sometimes I wonder if greed will take over, so that if a cure is found the Big Company(s) step in keep it from being released. Or has it already? Will the cure become like the epi-pen that only costs cents to make, but thousands to get a hold of, and some insurances won’t even cover it.
This is only the technical part of the disease. The physical part of the disease is a big money maker as well. What I mean is the food. It costs so much more to buy the healthy food. The only companies who have not seemed to try to make money off the diabetics are gum and J-ELLO. Hershey’s and any other company that has produced a sugar free snack, can only seem to do it at the expense of an arm and a leg. To buy a small bag of Hershey’s chocolate caramel candy bars–you get about 10 or less in a bag–it costs the same or less to buy a bag of mini-candy bars. We already pay an arm and a leg (or literally feet) just having the disease, why should we have to pay to feel not so left out. I realize that some might think that I AM being petty. Maybe I AM . But I have had Type 1 for 30 years. I was 14 when I was diagnosed. My youngest sister was diagnosed five years later, and she has been dead for the last 8 1/2 years due to the complications of the disease. I also have a cousin who was diagnosed a few years after my sister was.
With that being said, let’s focus on a CURE to put the money into, and not some gadget that may or may not help all of the diabetics out there. Yes, insulin saved a lot of children that were in comas, but with the all the modern research, we already know how to make our lives easier and to live longer. Eventually the disease just takes over, because the body can no longer keep the kidney’s from shutting down, the blood vessels in the eyes from popping, the circulation in the feet and legs from slowing down that results in nerve damage, or the heart just giving out. A CURE is where the money needs to really go. (I AM finished. Sorry for the novel and the soapbox I have been on since my sister passed away.)
So obviously this is not big news for me because its not really news. Apple has yet to announce anything close to replacing the test strip for diabetics. This is speculation and more hype for Apple as if pricking a finger is the worst thing a diabetic endures. Being type 2 for 15 years I can tell you, the last concern is having to check my glucose with a lancet and test strip. I’ve seen attempts before and much like the trouble with monitoring pulse through a wrist device. The ideal of a optical system able to accurate check every human’s glucose level is also something elusive to technology. Apple may indeed be working on a solution, but we may find the costs and procedures to get this approve through the FDA will be long and full of people who simply won’t benefit from it. Personally as a diabetic when I read the headlines some wrote about this. I really thought Apple was actually working on a solution to reverse diabetes and not simply another attempt at making a less evasive test for glucose. I know Steve Jobs died of pancreatic cancer and all of us diabetics want to live better lives. But Apple is getting far too much credit when credit is not do yet. This is just not big news that Apple is trying again where others have failed. Let’s talk about something when Apple actually has success.